La página a la que intenta acceder ha sido trasladada a otra dirección.
https://quibio.web.uah.es/
Por favor, espere a ser redirigido o pulse en el enlace anterior.
The page you are trying to access has been moved to another address.
https://quibio.web.uah.es/
Please wait to be redirected or click on the link above.
https://quibio.web.uah.es/group/
y actualice sus enlaces.
Publicaciones > Milián et al
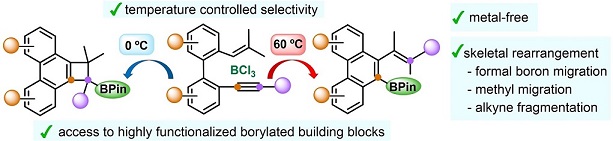
Metal-Free Temperature-Controlled Regiodivergent Borylative Cyclizations of Enynes: BCl(3) -Promoted Skeletal Rearrangement.
Universidad de Alcalá (IRYCIS). Departamento de Química Orgánica y Química Inorgánica, Instituto de Investigación Química "Andrés M. del Río" (IQAR). Campus Científico-Tecnológico, Facultad de Farmacia, Autovía A-II, Km 33.1, 28805-Alcalá de Henares, Madrid, Spain.
Keywords: Boron; Cyclization; Enynes; Phenanthrenes; Rearrangement
Abstract
Metal-free borylative cyclization of biphenyl-embedded 1,3,5-trien-7-ynes in the presence of simple and inexpensive BCl(3) provided synthetically useful borylated building blocks. The outcome of the process depends on the reaction temperature, with borylated phenanthrenes obtained at 60 °C and phenanthrene-fused borylated cyclobutanes formed at 0 °C. Based on DFT calculations, a mechanism for these novel transformations has been proposed, which involves an uncommon skeletal rearrangement, including migration of a methyl group and alkyne fragmentation, unprecedented in BCl(3) -promoted cyclization reactions.