XVII SIJ-RSEQ organisation

Several members of the group have been in charge of organising the 17th RSEQ Young Researchers Symposium, held in Alcalá de Henares from 23 to 26 November.
XVII Simposio de Investigadores Jóvenes de la RSEQ
The group has also made several contributions:
– Oral:
· Synthesis of seven- and eight-membered rings by Brønsted acid-catalyzed cyclization of biphenyl embedded trienynes
Jaime Tostado Sánchez, Juan J. Vaquero, Manuel A. Fernández-Rodríguez
– Flash:
· Enantio- and diastereoselective cyclopropanation of trans-alkenylboronates: Synthesis of versatile (trifluoromethyl)cyclopropylboronates
Julia Altarejos, David Sucunza, Juan José Vaquero, Javier Carreras
– Poster:
· Synthesis of a series of novel bicyclic amino acids
Álvaro González, Francisco José Torrero, José Luis Aceña, Juan José Vaquero
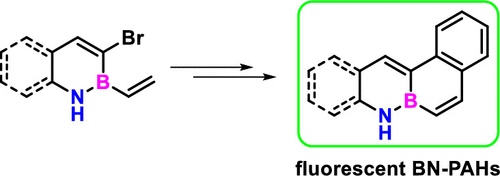
· Metal-free straightforward synthesis of boron-functionalized indenes and fulvenes by borylative cyclization
Ester Sans Panadés, Patricia García-García, Juan J. Vaquero, Cintia Virumbrales, Roberto Sanz, Manuel A. Fernández-Rodríguez
· SYNTHESIS OF PEPTIDE NUCLEIC ACIDS (PNAs) FOR THE TREATMENT OF CHRONIC KIDNEY DISEASE
Francisco Maqueda Zelaya, Verónica Miguel, José Luis Aceña, Santiago Lamas, Juan José Vaquero
· Synthesis of 4-membered rings fused lactams by gold(I) catalyzed
cyclization of alkynylcyclobutanamides
Guillermo G. Otárola, María Soledad Garre, Estíbaliz Merino, David Sucunza, Juan J. Vaquero, Patricia García-García
· Regioselective Ir-Catalyzed C−H Borylation of 4a,8a-Dihydro-4a-Aza-8a-Boranaphthalene
Isabel Valencia, Patricia García, David Sucunza, Juan J. Vaquero
· DEVELOPMENT OF A SYNTHETIC ROUTE TO LAETEVIRENOL A
Lucía Sánchez-Jiménez, Ana Milián, Jaime Tostado, Juan J. Vaquero, Patricia García-García, Manuel A. Fernández-Rodríguez
· Kinetic resolution in transannular Morita-Baylis-Hillman reaction:
approach to the synthesis of guaiane-type sesquiterpenes
Rubén Manzano, Raquel Mato, Efraím Reyes, Liher Prieto, Uxue Uria, Luisa Carrillo, Jose L. Vicario
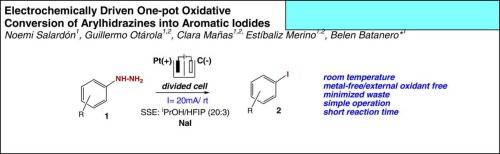
· Electrochemical synthesis of aryl iodides from arylhydrazines
Clara Mañas Hernández, Guillermo Otárola, Estíbaliz Merino, Noemi Salardón, Belén Batanero