Design and synthesis of bioactive compounds against targets involved in renal disease
Chronic kidney disease (CKD) is a major clinical and socioeconomic problem both globally and in Spain. Given its importance, multiple investigations are trying to deepen the understanding of the physiopathological basis of the disease, to try to identify high-risk populations, implement preventive measures and develop therapeutic strategies capable of alleviating the problem.
In this context, the search and validation of new targets and the strategies to activate or inhibit them using bioactive small molecules through medical chemistry in a multidisciplinary context is an objective of great interest.
Our research is focused on the design and synthesis of new inhibitors of Calpain, PTP1B (Protein Tyrosine Phosphatase 1B), NIK (NF-κB-Inducing Kinase) and ILK (Integrin-linked Kinase) trying to develop the hit-to-lead process, optimizations and candidate search in the preclinical phase.
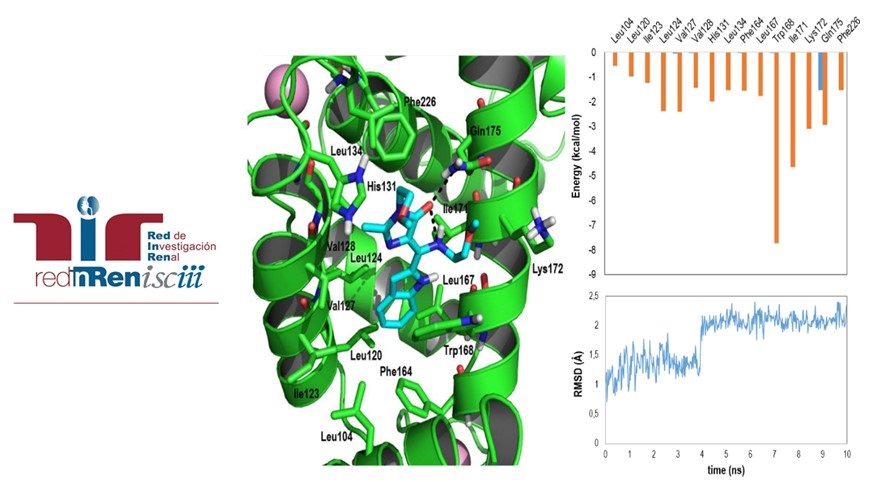
Docking and molecular dynamics of the calpain inhibitor MMG-1182
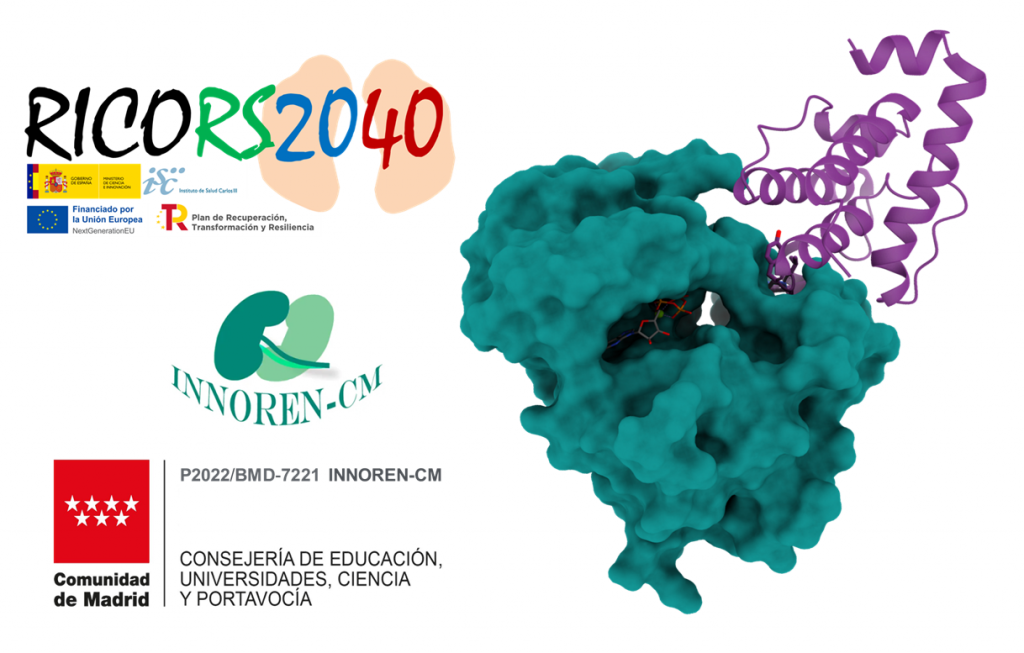
ILK-parvin protein-protein interaction
We also address novel preventive or curative strategies for CKD, studying the possibility of delivering high concentrations of drugs and small genetic modulators (miRNAs) to the kidney.
Recent publications:
Identification and study of new NF-κB-inducing kinase ligands derived from the imidazolone scaffold.
Francisco Maqueda-Zelaya, Lara Valiño-Rivas, Ana Milián, Sara Gutiérrez, José Luis Aceña, Javier Garcia-Marin, Mª Dolores Sánchez-Niño, Juan J Vaquero, Alberto Ortiz
Insight into the mechanism of molecular recognition between human Integrin-Linked Kinase and Cpd22 and its implication at atomic level.
Javier García-Marín, Diego Rodríguez-Puyol, Juan J Vaquero
RICORS2040: the need for collaborative research in chronic kidney disease.
Alberto Ortiz
A Computer-Driven Scaffold-Hopping Approach Generating New PTP1B Inhibitors from the Pyrrolo%1,2-a%quinoxaline Core.
Javier García-Marín, Mercedes Griera, Ramón Alajarín, Manuel Rodríguez-Puyol, Diego Rodríguez-Puyol, Juan J Vaquero
Pyrrolo%1,2-a%quinoxal-5-inium salts and 4,5-dihydropyrrolo%1,2-a%quinoxalines: Synthesis, activity and computational docking for protein tyrosine phosphatase 1B.
Patricia Sánchez-Alonso, Mercedes Griera, Javier García-Marín, Manuel Rodríguez-Puyol, Ramón Alajarín, Juan J Vaquero, Diego Rodríguez-Puyol
Tripeptides as Integrin-Linked Kinase Modulating Agents Based on a Protein-Protein Interaction with α-Parvin.
Javier Garcia-Marin, Mercedes Griera-Merino, Alejandra Matamoros-Recio, Sergio de Frutos, Manuel Rodríguez-Puyol, Ramón Alajarín, Juan J Vaquero, Diego Rodríguez-Puyol
The pHLIP system as a vehicle for microRNAs in the kidney.
Verónica Miguel, Carlos Rey, José Luis Aceña, Francisco Maqueda, Carlos Fernández-Hernando, Diego Rodríguez-Puyol, Juan J Vaquero, Santiago Lamas
NIK as a Druggable Mediator of Tissue Injury.
Lara Valino-Rivas, Juan Jose Vaquero, David Sucunza, Sara Gutierrez, Ana B Sanz, Manuel Fresno, Alberto Ortiz, Maria Dolores Sanchez-Nino
MAP3K kinases and kidney injury.
Leticia Cuarental, David Sucunza, Lara Valino-Rivas, Beatriz Fernandez-Fernandez, Ana Belen Sanz, Alberto Ortiz, Juan Jose Vaquero, Maria Dolores Sanchez-Nino
Discovery of potent calpain inhibitors based on the azolo-imidazolidenone.
Sara Gutierrez, Maria Moron, Mercedes Griera, David Sucunza, Laura Calleros, Andrea Garcia-Jerez, Claire Coderch, Francisco J Hermoso, Carolina Burgos, Manuel Rodriguez-Puyol, Beatriz de Pascual-Teresa, Maria L Diez-Marques, Antonio Jimenez, Miguel Toro-Londono, Diego Rodriguez-Puyol, Juan J Vaquero


