Esta versión de nuestra web ya no se mantiene actualizada.
Por favor, visite nuestra web operativa en
https://quibio.web.uah.es/group/
y actualice sus enlaces.
https://quibio.web.uah.es/group/
y actualice sus enlaces.
¡Gracias!
Synthesis of five-membered organoborate heterocycles via a metal-free carboboration and their use in cross-coupling reactions
Isabel Valencia, David Sucunza, Patricia García-García, Adrián Pérez-Redondo, Juan Vaquero López
Synthesis 2022, Accepted Articles
DOI: 10.1055/a-1979-6009
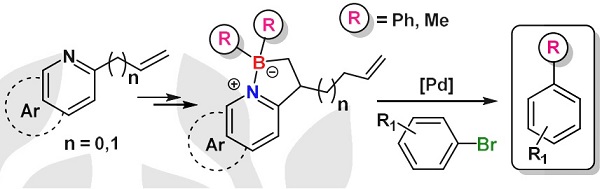
| Treatment of various vinylbenzopyridines with allyl(dichloro)borane affords five-membered organoborate heterocycles via a metal-free carboboration. The reaction between these organoborates and Grignard reagents increases the number of derivatives belonging to this novel family of four-coordinate organoboron compounds. Some of them were used as reagents in phenylations and methylations in moderate to high yields under standard palladium-catalyzed cross-coupling reaction conditions. |