https://quibio.web.uah.es/group/
y actualice sus enlaces.
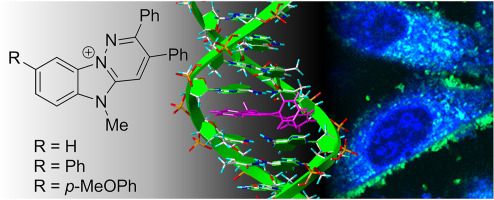
Publicaciones > Abengozar et al
Recent developments in the chemistry of BN-aromatic hydrocarbons.
Departamento de Química Orgánica y Química Inorgánica, Instituto de Investigación Química “Andrés M. del Río” (IQAR), Universidad de Alcalá (IRYCIS), Campus Científico-Tecnológico, Facultad de Farmacia, Alcalá de Henares, Madrid, Spain.
Abstract
Heterocycles containing both N and B heteroatoms in their structure were first reported by Dewar in the middle of the last century. However, they received little attention until the early years of this century, when several groups revisited these compounds due to their interest in BN/CC isosterism. As a result of these systematic studies, very significant advances have been made in our understanding of the chemistry of these BN-heterocycles. The purpose of this review is to summarize the most significant advances in the last two decades as regards the development of synthetic strategies and studies of their reactivity, as well as to provide an overview of their general properties and main applications.