Publications > García-García et al
Org Lett. 2026 Jan 23;28(3):907-911. doi: 10.1021/acs.orglett.5c04454. Epub 2025 Dec 3. PubMed 41338582
Synthesis of Multisubstituted Pyridines by Heterocyclization of TosMIC Derivatives: Total Synthesis of Caerulomycins A and K.
José A García-García, José Luis Aceña, Patricia García-García, David Sucunza
Departamento de Química Orgánica y Química Inorgánica, Instituto de Investigación Química "Andrés M. del Río" (IQAR), Universidad de Alcalá, IRYCIS, 28805 Alcalá de Henares, Madrid, Spain.
Abstract
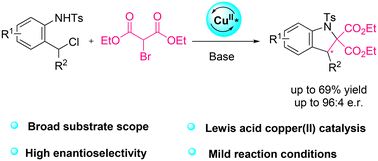
A concise synthesis of multisubstituted pyridines from α-allylic TosMIC derivatives and electrophiles is reported. The process involves a tandem heterocyclization exploiting the dual reactivity of the isocyanide group. The methodology tolerates various electrophiles, including halogen sources, and proceeds under mild conditions. Its utility is showcased in the total synthesis of caerulomycins A and K, in only five steps from TosMIC.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: