Alex Hipólito Barriuso
- FPI-UAH grant Universidad de Alcalá (2024-2028)
- Project associate researcher Universidad de Alcalá (2023)
- Master Drug Discovery, Universidad de Alcalá (2023)
- Graduated (Chemistry) at the Universidad de Alcalá (2022)

On April 4th, Clara Mañas successfully defended her doctoral thesis under the supervision of Dr. Estíbaliz Merino: “Design and study of novel reactivities of alkynylazobenzenes”
Thanks to all members of thesis tribunal: Dr. Teresa Quirós, Dr. Eva Maya and Dr. María Ribagorda
Congratulations to Dr. Mañas!

Yawen Hu, Cédric Hervieu, Dr. Estíbaliz Merino*, Prof. Cristina Nevado*
Angew. Chem. Int. Ed. 2024, Early View
DOI: anie.202319158
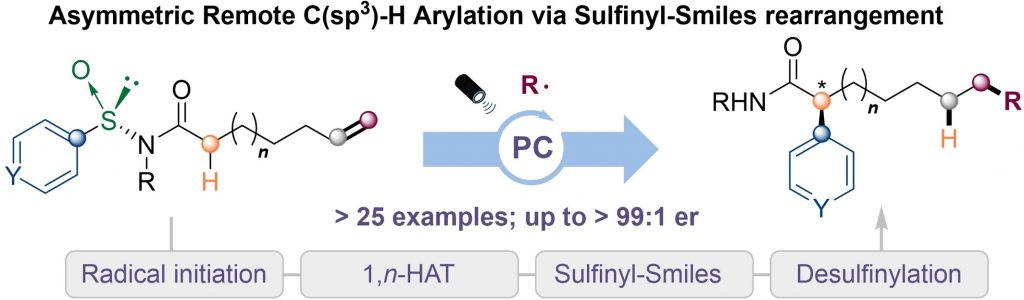
| An efficient asymmetric remote arylation of C(sp3)−H bonds under photoredox conditions is described here. The reaction features the addition radicals to a double bond followed by a site-selective radical translocation (1,n-hydrogen atom transfer) as well as a stereocontrolled aryl migration via sulfinyl-Smiles rearrangement furnishing a wide range of chiral α-arylated amides with up to >99 : 1 er. Mechanistic studies indicate that the sulfinamide group governs the stereochemistry of the product with the aryl migration being the rate determining step preceded by a kinetically favored 1,n-HAT process. |
Last friday, March 16th, Dr. García García was appointed as Full Professor at the UAH.
Congratulations Patricia!

Gold-Catalyzed Tandem Oxidation-Migration of 3-Propargyl Indoles: Synthesis of α-Indol-3-yl α,β-Unsaturated Carbonyls.
Palabras clave: carbonyls; gold; homogeneous catalysis; indoles; oxidation
Resumen
No hay resumen
Last friday, March 1st, Dr. Estibaliz Merino successfully passed her exam and was appointed as Associate Professor at the UAH.
Congratulations Estíbaliz!

Clara Mañas and Estíbaliz Merino*
Org. Lett. 2024, ASAP
DOI: acs.orglett.4c00097
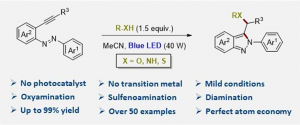
| We disclose the heterodifunctionalization of alkynylazobenzenes promoted exclusively by visible light in the absence of any transition metal and/or photocatalyst. This reaction features excellent regioselectivity on a broad variety of substrates with perfect atom economy. Alcohols, carboxylic acids, thiols, amides, heterocycles, and even water are suitable substrates for the promotion of the oxyamination, sulfenoamination, and diamination reactions. In this manner, biologically active indazole scaffolds can be rapidly assembled from alkyne feedstocks. |

Assistant Professor

